Artificial intelligence will design cranial implants not only equally good as humans, but significantly faster – envisions Dr Eng. Marek Wodziński from the Faculty of Electrical Engineering, Automatics, Computer Science, and Biomedical Engineering. Tens of thousands of people can benefit from this project.
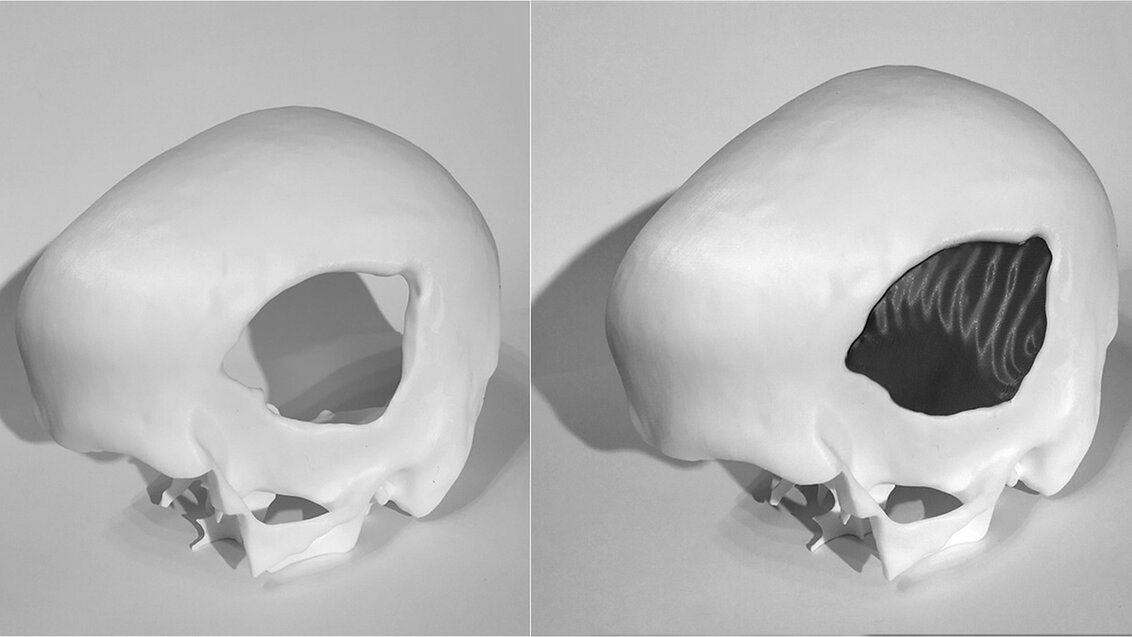
To fill a defect, all you have to do is design an implant that can replace the damaged bone. Then, using a material approved for medical use, manufacture its physical form and finally perform the replacement surgery. In the majority of cases, medicine sees no obstacles in successfully carrying out such surgeries – science gave us all the tools we need to help people with cranial defects.
However, in 2019, people in need of a cranial implant amounted to about 5 million patients around the world who, despite medical recommendations, had not undergone a neurosurgical procedure that would allow them to recover completely. Similarly, in Poland, many people who had been deprived of an intact mechanical barrier of the brain as a result of surgical intervention due to injury or cancer are forced to wait for an implant for months. During the wait, places and situations that for most of us are merely everyday nuisances can pose fatal threats to them. Moreover, such patients also struggle with social exclusion or reluctance to socialise because they feel different due to deformations. This long waiting time for a surgery has two main reasons – the process of manufacturing personalised cranial implants is currently extremely time-consuming and the number of neurosurgeons is insufficient.
Swiftness requires innovation
Implants are designed by bioengineers who are qualified to work with specialised software. Modelling an implant optimally adjusted to the defect in question requires them to individually analyse the medical data of a patient – this can take from several to several dozen hours. To make the design a reality, the project is usually sent to another company that produces the implant using, for instance, 3D printers; and then to yet another company that will sterilise it (if this procedure is not performed in a hospital). The entire process of approving the use of the implant can be considerably prolonged and take weeks or even months.
The AGH UST scientist, Dr Eng. Marek Wodziński, noticed the problem and decided to have a go at solving it. The National Centre for Research and Development, within the LIDER competition, rewarded his research project titled: Innowacyjny system do projektowania i weryfikacji spersonalizowanych implantów czaszkowych oparty o sztuczną inteligencję i mieszaną rzeczywistość [Innovative system for designing and verifying personalised cranial implants based on artificial intelligence and mixed reality]. The young scientist received PLN 1,500,000 to implement the project.
‘The innovation is that the system will automatically design a personalised implant for a given patient. Immediately in the operating theatre, a neurosurgeon will receive a tailor-made model ready to be printed by pushing a single button’, the project leader says about his goals.
Dr Marek Wodziński can already boast about a success related to a research activity in the medical industry – last year, he received the first prize in the ABB competition for his doctoral dissertation titled: Medical image registration methods focused on the problem of missing data, in which he proposed a series of algorithms aimed at increasing the quality of medical imaging and as direct support for medical procedures. Furthermore, for his research activity, he was awarded the START scholarship, granted by the Foundation for Polish Science, and a ministerial scholarship for exceptional young scientists.
Deeper insight brings a new quality
‘We’ve already designed algorithms that can successfully fill a cranial defect. The problem is that very often the cranial defect itself is not compatible with the implant, as the former can have a shape that physically won’t fit in the cranial cavity’, Dr Marek Wodziński says about the difficulties related to programming this innovative method. ‘Sometimes, as a result of traffic accidents, the skull is first damaged and then broken, which renders the situation more dangerous on the inside than on the outside – and we can’t put something in from the inside of the skull. Therefore, the replacement procedure requires further modifications to the defect model. Additionally, implants are made of different materials. So, depending on the material favoured by a given neurosurgeon, the implant will yield different mechanical and different biological responses – this must also be taken into account during the design stage. Some implants will have to be thicker, others thinner; they are also fitted in various ways. We must also consider the fact that immediately after the procedure the brain can be deformed and we may have to deal with swelling. Implants should not cause an increase in intracranial pressure; therefore, they should always be a bit thinner than the original cranium. There’s still a long way to go from the defect to the implant’, explains the scientist.
Previous attempts at creating algorithms to design implants by other research teams have yielded moderate results. These algorithms can deal quite well with designing implants for defects similar to those they had analysed before, but cannot transpose their expertise to new cases with which they had not dealt before.
‘The project will definitely have to account for increasing the ability to generalise these proposed algorithms to include cases that had not been shown to the software during training’, says the project leader.
 14th Krakow Integration Days
14th Krakow Integration Days  Professor Wojciech Górecki – AGH UST Honorary Professor
Professor Wojciech Górecki – AGH UST Honorary Professor  Thermal treatment of municipal and biomass wastes. Research of AGH UST scientists
Thermal treatment of municipal and biomass wastes. Research of AGH UST scientists  Professor Janusz Mroczka – AGH UST Doctor Honoris Causa
Professor Janusz Mroczka – AGH UST Doctor Honoris Causa